Empatica wearable gets CE mark for detecting Covid-19
- March 15, 2021
- Steve Rogerson
MIT spinoff Empatica has become the first to receive a European CE mark for detecting early symptoms of Covid-19 using wearables.
The Aura wearable platform can automatically provide an early warning of a possible respiratory infection through Empatica’s clinically validated algorithm.
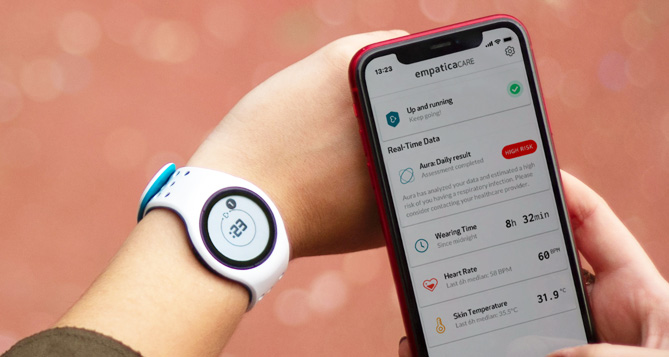
Digital biomarker and medical device company Empatica’s Aura system can detect and alert to respiratory infections, including Covid-19, early and before visible symptoms appear. It received its CE mark as a Class lla medical device, meaning that Aura, as well as Empatica’s remote health monitoring platform, Care, meet safety and performance requirements in Europe. This makes Aura the first product of its kind to receive CE-marking for respiratory infection detection using smart wearable technology.
Aura’s algorithm analyses vital signs from Empatica’s smartwatches, comparing data against historical baselines unique to each wearer. Requiring no manual input, when changes in a user’s data suggest their body is beginning to fight an infection, Aura automatically displays a warning to Empatica’s smartphone-based Care app, as well as to the cloud-based Care Portal, optionally informing their healthcare provider or caregivers.
Through the Care Portal, healthcare professionals can simultaneously monitor the risk status of hundreds of individuals remotely, making Aura a tool for maintaining healthy communities and preventing outbreaks.
In validation studies, Aura showed a sensitivity of 0.94 for detecting patients with a possible infection. Detection happened on average two days after viral inoculation. The results apply to general physiological responses that arose from three different viruses – H1N1, Rhinovirus and Sars-CoV-2.
Covid-19 already has resulted in millions of deaths globally, while other respiratory infections caused by influenza-like pathogens kill between 250,000 and 500,000 people and cost up to $167bn annually.
“Our goal with Aura was to create an effective, scalable, and affordable tool for screening infection risk and supporting testing efficiency, all while monitoring people’s health remotely,” said Empatica CEO Matteo Lai. “This result is a win for science and technology against the Covid-19 pandemic. We hope Aura can play an important role in controlling new outbreaks and helping people safely get back to work and to a normal life.”
Aura, for use in ages 14 and up, is now available across Europe for healthcare providers and other professionals seeking to contain the spread of infectious diseases within their communities.
Empatica, in the process of gaining FDA-approval for Aura, has partnered with the US Department of Health & Human Services via the Biomedical Advanced Research & Development Authority and Stanford University to run a nationwide validation study with US-based healthcare workers.
Empatica, an MIT-spinoff based in Boston, Massachusetts, and Milan, Italy, is a pioneer in digital biomarker development and continuous patient monitoring driven by AI. Its smartwatches are FDA cleared and CE-marked, and have been sold to thousands of institutional partners for clinical trials and research, in studies examining depression, addiction, stress, oncology, epilepsy, migraine and other conditions.





